Introduction
For any food brand selling in the United States, FDA compliance isn’t optional — it’s the baseline. The FDA sets specific requirements for how food products have to be labeled, and missing even one of them can put your product at risk of being pulled from shelves or flagged by a retailer before it ever gets there.
This post covers the specific FDA compliance requirements that apply to food packaging labels, what each one actually means in practice, and where brands most commonly run into trouble. If you’ve already read our broader label compliance guide, consider this the deeper dive on the food side.
FDA Compliance and the Nutrition Facts Panel
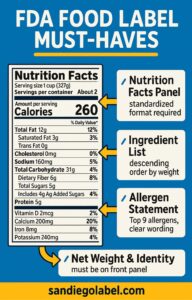
The Nutrition Facts panel is probably the most recognized element on any food label, and it’s also one of the most tightly regulated. FDA compliance here doesn’t just mean including nutritional information — it means presenting that information exactly the way the FDA specifies.
That means standardized formatting for font sizes, type weight, line spacing, and the order nutrients are listed. Serving size declarations follow their own set of rules, including reference amounts customarily consumed (RACC) that the FDA has established by product category. You can’t decide that your serving size is whatever makes your product look best nutritionally. The serving size has to reflect how people actually eat the product.
The two most common Nutrition Facts mistakes are using a font that falls below the minimum size requirement, and formatting the panel based on what looks good in the design software instead of what the FDA template actually specifies. Both are easy to miss in a design review and both will get flagged.
FDA Compliance and the Ingredient List
Every ingredient in your product has to be listed, and they have to appear in descending order by weight. The heaviest ingredient goes first. This applies to sub-ingredients too — if you’re using a compound ingredient like salsa, you either list its components individually in order of weight, or you list the compound ingredient with its components in parentheses immediately after.
The FDA also requires that you use common or usual names for ingredients. “Sugar” instead of “sucrose.” “Salt” instead of “sodium chloride.” The goal is that a consumer reading the label can understand what’s in the product without a chemistry background.
One area that trips up smaller brands: if you’re using a flavoring, you don’t have to list every component of that flavor, but you do have to declare it as “natural flavor,” “artificial flavor,” or both, depending on its composition. Getting that distinction wrong is an FDA compliance issue.
FDA Compliance and Allergen Labeling Under FALCPA
The Food Allergen Labeling and Consumer Protection Act requires clear declaration of the nine major allergens recognized by the FDA: milk, eggs, fish, shellfish, tree nuts, peanuts, wheat, soybeans, and sesame.
There are two acceptable ways to declare allergens. The first is to include the allergen name in parentheses within the ingredient list itself — for example, listing “casein (milk)” so the connection is explicit. The second is a separate “Contains” statement after the ingredient list, such as “Contains: Milk, Wheat.”
A few things worth knowing. If you use a “Contains” statement, it has to be complete. You can’t list some allergens in the ingredient list and others in the statement — if you use it, it has to reflect all allergens present. Advisory statements like “may contain traces of peanuts” are voluntary and don’t substitute for mandatory allergen declarations. If the allergen is actually in the product, it has to be declared.
Net Quantity and Statement of Identity
Both of these have to appear on the principal display panel, which is the front-facing surface of your package — the side a consumer sees first on a shelf.
The statement of identity is simply what the product is. “Salsa,” “Organic Whole Milk,” “Roasted Almonds.” It has to be prominent, in a type size reasonably related to the most prominent printed matter on the panel, and it has to use the common or usual name of the product.
The net quantity declaration tells the consumer how much product is in the package. It has to appear in the bottom 30% of the principal display panel and be expressed in both U.S. customary and metric units for most product types. Font size minimums here are calculated based on the total area of the principal display panel, so the right size depends on your packaging dimensions.
Placement, Readability, and FDA Compliance Design Rules
The FDA distinguishes between the principal display panel and the information panel, which is typically the side or back of the package. The Nutrition Facts panel, ingredient list, allergen declarations, and manufacturer information generally belong on the information panel, while the statement of identity and net quantity go on the principal display panel.
A few design-side details that matter for FDA compliance:
The information panel has to be the label panel immediately to the right of the principal display panel as the consumer faces it. Placing required information elsewhere — even if it’s technically on the label — can be a compliance issue.
Minimum type sizes apply based on package size and the specific information being displayed. A font that reads fine on a standard bottle may fall below the minimum for net quantity on a small-format package.
Labels on curved or irregular surfaces need to maintain legibility after application. A proof that looks compliant flat doesn’t always stay that way once it’s applied to a round container.
Common FDA Compliance Mistakes
A few patterns come up repeatedly:
Formatting the Nutrition Facts by eye instead of by template. The FDA provides standardized formats for a reason. Using them as a starting point prevents most formatting errors.
Allergen declarations that are present but incomplete. A “Contains” statement that lists milk but misses wheat because wheat only appears as a sub-ingredient is still non-compliant.
Net quantity font size calculated incorrectly. This one is easy to miss because it’s tied to packaging dimensions, not a fixed size. If your package size changes between versions, the minimum font size may change too.
Ingredient list split across panels. The ingredient list has to appear as a single, continuous block. Breaking it across two panels to make the design work is not compliant.
FDA compliance review happening too late. The most expensive version of any of these mistakes is discovering them after a run is printed. Compliance review should be a gate in your approval process, not a final check.
How San Diego Label Approaches This
We work with food brands at different stages — some are launching a first product and building their label from scratch, others are updating packaging and want to make sure the new version meets current FDA compliance requirements. In both cases, we review files for compliance issues before anything goes to press.
That’s not a guarantee of legal compliance — we’re printers, not regulatory attorneys — but catching a formatting error or a missing declaration before a run prints is a lot less painful than catching it after. If you’re building a new food label or updating an existing one and want a second set of eyes before you commit to a run, reach out here.
